Digester bio-gas to bio-methanol
The complete biological decomposition of organic matter to methane (CH4) and carbon dioxide (CO2) under oxygen-depleted conditions – i.e. anaerobic – is complicated and is an interaction between a number of different bacteria that are each responsible for their part of the task. What may be a waste product from some bacteria could be a substrate (or food) for others, and in this way the bacteria are interdependent. Compared with the aerobic (oxygen-rich) decomposition of organic matter, the energy yield of the anaerobic process is far smaller. The decomposition of, for example, glucose will under aerobic conditions give a net yield of 38 ATP molecules, while anaerobic decomposition will yield only 2 ATP molecules. This means that the growth rate of anaerobic bacteria is considerably lower than that of aerobic bacteria and that the production of biomass (in the form of living bacteria) is less per gram decomposed organic matter. Where aerobic decomposition of 1 g substance results in the production of 0.5 g biomass, the yield under anaerobic conditions is only 0,1 g biomass.

The biogas process is often divided into three steps: Hydrolysis, acidogenesis and methanogenesis, where different groups of bacteria are each responsible for a step (see figure 1).
Figure 1: The anaerobic decomposition of organic matter consists of three main phases: A. Hydrolysis (1a, 1b, 1c). B. Acidogenesis, also called fermentation (2, 3, 4). C. Methanogenesis (5, 6).
Methanol is produced from synthesis gas, which is normally produced from raw material – natural gas or biogas. A compari-son of methanol production from two varieties of raw materials – were made – natural gas and biogas.
The basic starting point for comparison is the same mass inlet flow rates for both raw materials under the same operating conditions. Methanol production using natural gas and biogas as the raw materials was simulated using an Aspen Plus simula-tor with real chemical thermodynamic, and 16 146 kg/h crude methanol from natural gas and 14 615 kg/h from biogas could be produced. Methanol production from biogas could also in-crease by 9.7 % with processed operational and parametric modification using nonlinear programming (NLP).
The NLP model contains equations which enable methanol and steam production, and parametric optimization. The most im-portant is the conversion of methane in the reformer. Optimal methane conversion could take place by operating by the use of optimal parametric data in a reformer unit. Mathematical problems could include equations which present synthesis gas composition, crude methanol, and steam productions. The pri-mary objective of retrofit is to change the raw material with a minimum of additional cost and maximize the production of methanol and steam.
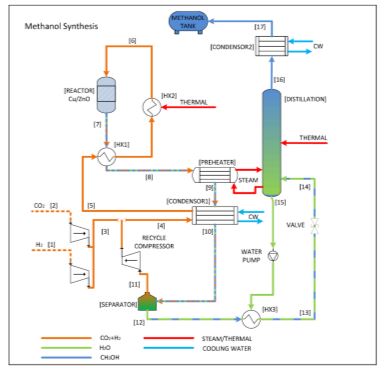
The optimal production of methanol was 16 040 kg/h under op-timal parameters (temperature=840° C and pressure=8 bar) in the reformer. Optimal steam production was 13 230 kW. The total methanol mass flow from biogas under optimal conditions was identical with the total methanol mass from natural gas.
Figure 2: Traditional methanol synthesis.